The science behind Qbtesting
Explore the clinical evidence supporting our
objective ADHD tests
Our tests involve motion tracking systems and computerized tasks that require continuous attention and impulse control. As a result, you are provided with data on core ADHD symptoms of hyperactivity, impulsivity, and inattention.
Our technology can be used in children (6–12 years) and adolescents and adults (12–60 years). The computerized tasks differ in cognitive demand between the child version (go/no-go paradigm) and the adolescent/adult version (unconditional identical pair paradigm).
To get detailed information about our objective technology, connect with our expert clinical advisors.
Disclaimer: QbTest and QbCheck are considered equivalent in their clinical utility, intended use, and ability to measure the core symptoms of ADHD. For further information, visit our legal page.

1000000+
test takers benefited
35,000+
clinical users worldwide
20+
years of research
18
countries served
What the evidence shows
Objective measurement of hyperactivity, inattention, and impulsivity in a single assessment
Increased diagnostic accuracy when combined with clinical interviews and rating scales
Reduction in time to diagnosis and improved clinician confidence in decision-making
Ability to detect treatment response within hours of pharmacological interventional
Sensitivity to long-term symptom changes across different patient populations and treatments
Comparable validity and reliability in virtual and in-clinic settings
What the evidence shows
Objective measurement of hyperactivity, inattention, and impulsivity in a single assessment
Increased diagnostic accuracy when combined with clinical interviews and rating scales
Reduction in time to diagnosis and improved clinician confidence in decision-making
Ability to detect treatment response within hours of pharmacological interventional
Sensitivity to long-term symptom changes across different patient populations and treatments
Comparable validity and reliability in virtual and in-clinic settings
Our medical experts
Our clinical research team brings together decades of experience across medical and clinical science, and digital health. Their expertise spans clinical trials, advanced neuroimaging, and digital mental health research. With backgrounds in pharmaceutical and medical device research, they understand the importance of clinical rigor, regulatory standards, and real-world impact.
Their work centers on advancing ADHD care towards a more objective, data-driven future, giving you tools that are grounded in evidence. By bringing evidence-based thinking into innovative technologies, they help close the gap between research and day-to-day patient care.
Behind the science, you’ll find a team focused on one thing: helping you make more confident clinical decisions.

Medical research publications

Objective ADHD improvements align with patient-reported outcomes following remote treatment workflow (2025)

Objective assessment of attention deficit hyperactivity disorder with QbMobile: A smartphone application for clinical use (2025)
Medical research articles

Objective ADHD improvements align with patient-reported outcomes following remote treatment workflow (2025)

Objective assessment of attention deficit hyperactivity disorder with QbMobile: A smartphone application for clinical use (2025)
Clinical evidence supporting objective ADHD
assessment and treatment
Diagnostic accuracy and support
A randomized controlled study (Hollis et al., 2018) showed that clinicians with access to QbTest results were 44% more likely to make a diagnostic decision within a 6-month period. At 6 months, 76% of patients had received a diagnosis compared to 60% of the control group.
QbTest reduced appointment length by 15%, and clinicians were twice as likely to exclude a diagnosis of ADHD. They reported being more confident in their diagnostic decision, without compromising diagnostic accuracy.
QbTest had a stand-alone accuracy of 79% in adults and 78% in children. This accuracy increased to 89.5% in adults and 86.7% in children when combined with subjective measures. (Emser et al., 2018).

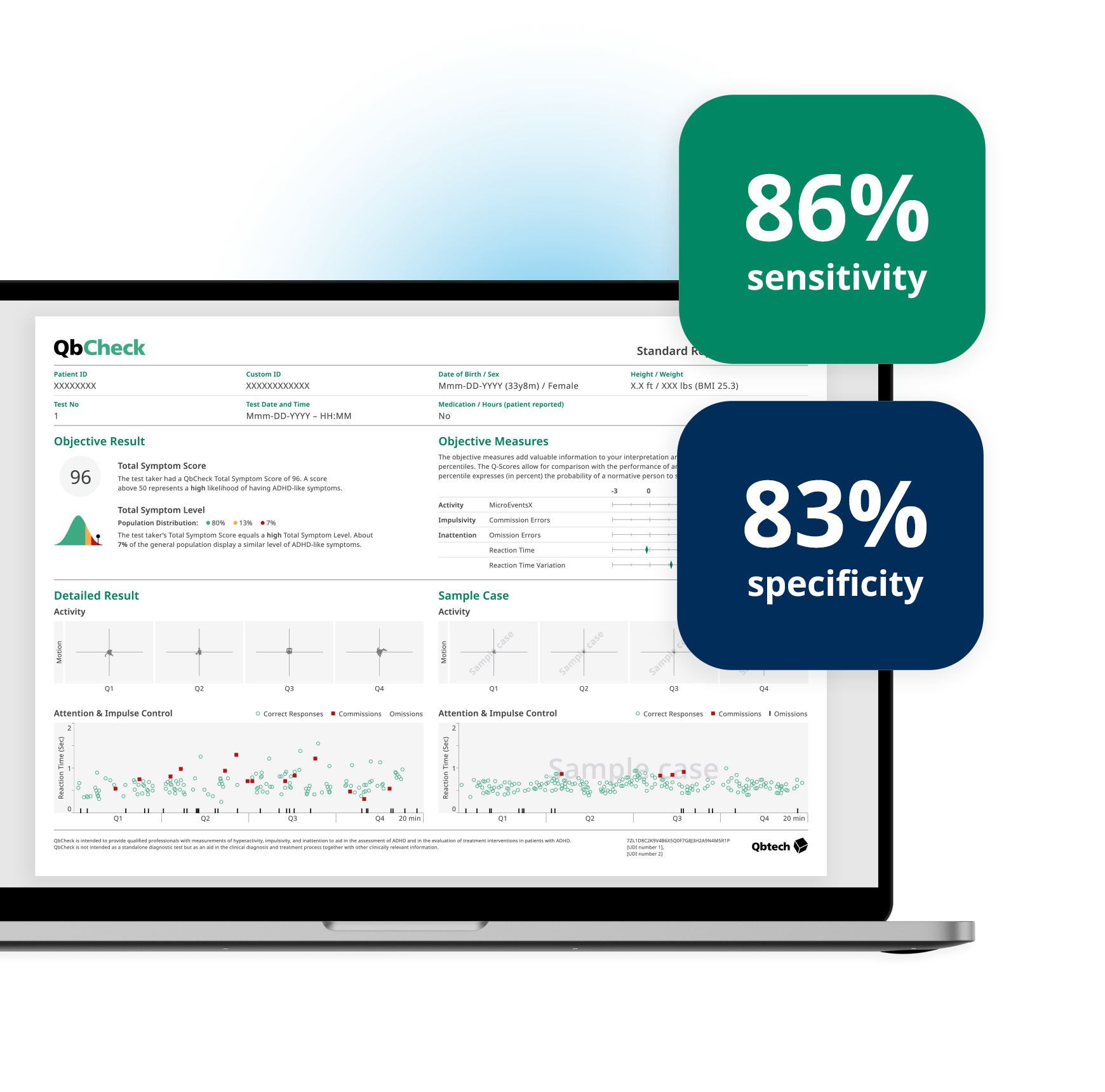
Validity and reliability
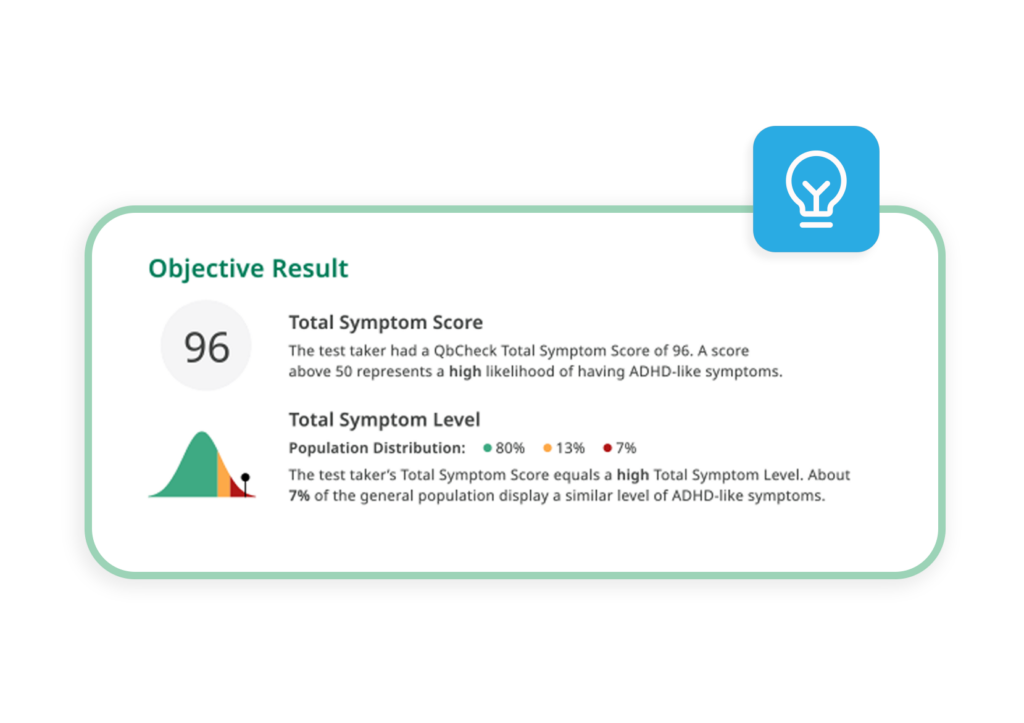
A composite measure based on hyperactivity, inattention, and impulsivity yielded 86% sensitivity and 83% specificity (Edebol et al., 2013a).
QbCheck demonstrated good test–retest reliability, good diagnostic validity, and equivalence to QbTest in total symptom scoring (Ulberstad et al., 2020).
A high test–retest reliability of QbTest has been found after medication treatment intervention (Löhman et al., 2023).
Treatment response and monitoring
QbCheck demonstrated significant improvement in symptom measures following treatment, including a reduction in total symptom score by 42% (Sanyal et al., 2024).
QbTest can distinguish medication treatment effect within hours of pharmacological titration and can also be used as an aid for evaluation of long term-term treatment (>1 year) of ADHD (Gustafsson and Hansen 2023b)
QbTest was able to identify symptom level reduction across dose levels, including cases where subjective rating scales did not detect improvement (Edebol et al., 2013b).

Broader clinical insights
QbTest has been shown to differentiate ADHD from normative individuals and from other clinical groups, including Bipolar II disorder, borderline personality disorder, and autism spectrum disorder (Edebol et al., 2012; Groom et al., 2016).
Improvements in QbTest scores have been correlated with improvements in quality of life and daily functioning (Martin-Key et al., 2022).
QbTest as part of the diagnostic workup for attention deficit hyperactivity disorder in children (age < 18 years), when used in combination with clinical assessment, may be cost-effective (Tomlinson et al., 2025).
Featured clinical studies

More than 50 studies using QbCheck and QbTest have been published,
the majority of which are peer-reviewed independent publications.
FAQs
Can Qbtesting support differential diagnosis?
A comprehensive review of ten studies determined a pooled sensitivity of 0.84 and a specificity of 0.84 for QbTest. The authors concluded this demonstrated QbTest’s ability to discriminate across a mixed population of normative and differential diagnosed individuals. (Gustafsson & Hansen, 2023)
Can digital ADHD tests be used in complex cases?
Clinicians in a childhood randomized controlled trial found value in using QbTest for more complex patient cases, which included comorbidities. QbTest was considered particularly helpful in cases where there was contention about treatment approaches. (Williams et al., 2021).
Can QbTest and QbCheck be used for diagnosing ADHD in older adults?
QbCheck and QbTest are for use in children (6-12 years) and in adolescents/adults (12-60 years). However, a study investigated the use of QbTest with adult ADHD patients aged 55-79. QbTest had a stand-alone accuracy of 70%, which increased to 91 % when combined with self-reports of ADHD severity (Bijlenga et al., 2019).
Do patients find the results from Qbtesting useful?
Survey data suggests that clinicians, service users, and their families find QbTest helpful and acceptable (Pellegrini et al., 2020). In another study, it was found that QbTest helped patients understand their symptoms and the diagnostic decision. (Hall et al., 2024)
Can QbCheck be used remotely?
In a study where QbCheck was used for remote monitoring of ADHD medication treatment a significant improvement (p<0.001) from baseline to post-treatment follow-up was seen in all five parameters (microevent, commission error, omission error, reaction time and reaction time variance). Total Symptom Score also reduced by 42% following starting medication. The authors concluded that QbCheck is a useful objective measure that could be incorporated in guiding treatment decisions, remote monitoring of ADHD medication, tracking of ADHD symptom regulation, and optimizing treatment outcomes for those with ADHD. (Sanyal et al., 2024).
References and sources
Bijlenga, D., Ulberstad, F., Thorell, L. B., Christiansen, H., Hirsch, O., & Kooij, J. J. S. (2019). Objective assessment of attention-deficit/hyperactivity disorder in older adults compared with controls using the QbTest. International Journal of Geriatric Psychiatry, 34(10), 1526–1533. https://doi.org/10.1002/gps.5163
Edebol, H., Helldin, L., & Norlander, T. (2012). Objective Measures of Behavior Manifestations in Adult ADHD and Differentiation from Participants with Bipolar II Disorder, Borderline Personality Disorder, Participants with Disconfirmed ADHD as Well as Normative Participants. Clinical Practice and Epidemiology in Mental Health: CP & EMH, 8, 134–143. https://doi.org/10.2174/1745017901208010134
Edebol, H., Helldin, L., & Norlander, T. (2013a). Measuring adult Attention Deficit Hyperactivity Disorder using the Quantified Behavior Test Plus. PsyCh Journal, 2(1), 48–62. https://doi.org/10.1002/pchj.17
Edebol, H., Helldin, L., & Norlander, T. (2013b). The weighed core symptom scale and prediction of ADHD in adults—Objective measures of remission and response to treatment with methylphenidate. Clinical Practice and Epidemiology in Mental Health: CP & EMH, 9, 171–179 https://doi.org/10.2174/1745017901309010171
Emser, T. S., Johnston, B. A., Steele, J. D., Kooij, S., Thorell, L., & Christiansen, H. (2018). Assessing ADHD symptoms in children and adults: Evaluating the role of objective measures. Behavioral and Brain Functions: BBF, 14(1), 11. https://doi.org/10.1186/s12993-018-0143-x
Groom, M. J., Young, Z., Hall, C. L., Gillott, A., & Hollis, C. (2016). The incremental validity of a computerised assessment added to clinical rating scales to differentiate adult ADHD from autism spectrum disorder. Psychiatry Research, 243, 168–173. https://doi.org/10.1016/j.psychres.2016.06.042
Gustafsson, U., & Hansen, M. (2023a). QbTest in the clinical assessment of attention deficit hyperactivity disorder: A review of the evidence. Mental Health Science. https://doi.org/10.1002/mhs2.43
Gustafsson, U., & Hansen, M. (2023b). QbTest for Monitoring Medication Treatment Response in ADHD: A Systematic Review. Clinical Practice and Epidemiology in Mental Health, 19(1). https://doi.org/10.2174/0117450179276630231030093814
Gustafsson, U., Larsson, S., Casals, N., Nolen, R., Sanyal, R. Y., & Hansen, M. (2025). Objective assessment of attention deficit hyperactivity disorder with QbMobile: A smartphone application for clinical use. Clinical Practice & Epidemiology in Mental Health, 21, e17450179444324. https://doi.org/10.2174/0117450179444324251118134538
Hall, S. S., McKenzie, C., Thomson, L., Ingall, B.-R., Groom, M. J., McGlennon, N., Dines-Allen, M., & Hall, C. L. (2024). A national evaluation of QbTest to support ADHD assessment: A real-world, mixed methods approach. BMC Health Services Research, 24(1), 1201.https://doi.org/10.1186/s12913-024-11693-7
Hollis, C., Hall, C. L., Guo, B., James, M., Boadu, J., Groom, M. J., Brown, N., Kaylor-Hughes, C., Moldavsky, M., Valentine, A. Z., Walker, G. M., Daley, D., Sayal, K., Morriss, R., & the AQUA Trial Group. (2018). The impact of a computerised test of attention and activity (QbTest) on diagnostic decision-making in children and young people with suspected attention deficit hyperactivity disorder: Single-blind randomised controlled trial. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 59(12), 1298–1308. https://doi.org/10.1111/jcpp.12921
Löhman, M., Domingo, B., Östlund, M., & Jansson, L. (2023). Contrasting expectancy effects with objective measures in adults with untreated ADHD during QBTEST. Scandinavian Journal of Psychology, sjop.12906. https://doi.org/10.1111/sjop.12906
Martin-Key, N. A., Stevenson, A., & Roy, P. (2022). Investigating the Clinical Utility of the Combined Use of Objective and Subjective Measures of ADHD During Treatment Optimization. Journal of Clinical Psychopharmacology, 42(2), 146–153. https://doi.org/10.1097/JCP.0000000000001350
Nolen, R., Sanyal, R., Larsson, S., Casals, N., Murdoch, T., Whelan, R., Hansen, M., & Anderton, P. (2025). Objective ADHD improvements align with patient-reported outcomes following remote treatment pathway. Qbtech. https://www.qbtech.com/blog/remote-objective-adhd-assessment-to-measure-quality_of_life_outcomes-2
Pellegrini, S., Murphy, M., & Lovett, E. (2020). The QbTest for ADHD assessment: Impact and implementation in Child and Adolescent Mental Health Services. Children and Youth Services Review, 114, 105032. https://doi.org/10.1016/j.childyouth.2020.105032
Sanyal, R., Nolen, R., Gustafsson, U., & Hansen, M. (2024). Utilizing Remote Objective ADHD Testing to Monitor Symptom Improvement Following Medication Treatment. International Journal of Psychiatry Research, 7(3). https://doi.org/10.33425/2641-4317.1195
Tomlinson, E., Ward, M., Walker, J., Benevente, M., Wang, H., Cooper, C., Jones, H. E., Owen-Smith, A., Lopez Manzano, C., James, S., Hank, D., Welton, N. J., & Whiting, P. (2025). Clinical and cost-effectiveness of technologies for the assessment of attention deficit hyperactivity disorder: A systematic review and economic model. Health Technology Assessment, 1–216. https://doi.org/10.3310/DRDR7171
Ulberstad, F., Boström, H., Chavanon, M.-L., Knollmann, M., Wiley, J., Christiansen, H., & Thorell, L. B. (2020). Objective measurement of attention deficit hyperactivity disorder symptoms outside the clinic using the QbCheck: Reliability and validity. International Journal of Methods in Psychiatric Research, 29(2), e1822. https://doi.org/10.1002/mpr.1822
Williams, L., Hall, C. L., Brown, S., Guo, B., James, M., Franceschini, M., Clarke, J., Selby, K., Vijayan, H., Kulkarni, N., Brown, N., Sayal, K., Hollis, C., & Groom, M. J. (2021). Optimising medication management in children and young people with ADHD using a computerised test (QbTest): A feasibility randomised controlled trial. Pilot and Feasibility Studies, 7(1), 68. https://doi.org/10.1186/s40814-021-00788-1
